
Hey guys, welcome to this Mometrix video on macromolecules. In our last video we took a look at cellular function, well macromolecules are the biological molecules that make cellular structure and function possible.
In this video we will take a look at the structure, the function, and source of the four macromolecules: carbohydrates, proteins, lipids(fats), and nucleic acids.
Carbohydrates
Structure
Carbohydrates are made up of carbon, hydrogen, and oxygen. There are three classes of carbohydrates: monosaccharides, disaccharides, and polysaccharides.
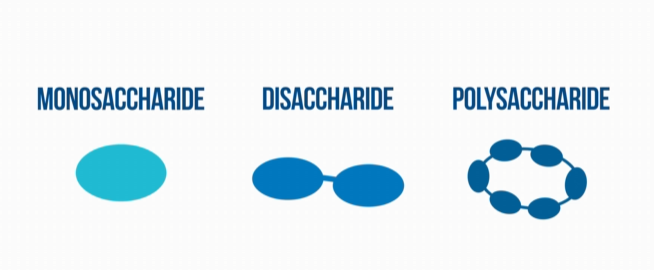
Monosaccharides are the most basic form of a carbohydrate. Since they are the most basic form of carbohydrates, they cannot be further broken down into simpler sugars. They are distinguished by the amount of carbon atoms they contain; which is between 3, and, rarely more than, 6 carbon atoms. Monosaccharides are white, crystalline solids. Monosaccharides usable for living organisms include: glucose (the most common), galactose, and fructose.
Disaccharides are made up of two monosaccharides. Examples of disaccharides include: sucrose, maltose, and lactose. Polysaccharides are composed of long chains of monosaccharides as the name suggests. Examples of polysaccharides include: starch and glycogen. How a carbohydrate is structured determines how energy is stored, and just how the breaking of carbohydrate molecular bonds let off energy.
Function
Carbohydrates supply our bodies with readily usable cellular energy. Glucose, the simplest carbohydrate, enters into our cells’ mitochondria, is converted into energy in the form of ATP and is used for various cellular functions. Carbohydrates are also used to tag lipids and proteins which helps in immune function, cellular communication, and allow for cells to attach to the necessary extracellular material. Our central nervous system and muscles need a lot of energy in order to function, and this energy comes from carbohydrates.
Source
Consumable carbohydrates can be found naturally occurring in fruits, vegetables, milk products, and grains.
Proteins
Structure
Proteins are the most structurally complex biological molecules. Proteins are made up of chains of polypeptides (which are made up of amino acids that are connected together by peptide bonds). There are three types of proteins: fibrous proteins (which are structural), globular proteins (which are functional proteins), and membrane proteins (which are interactive).
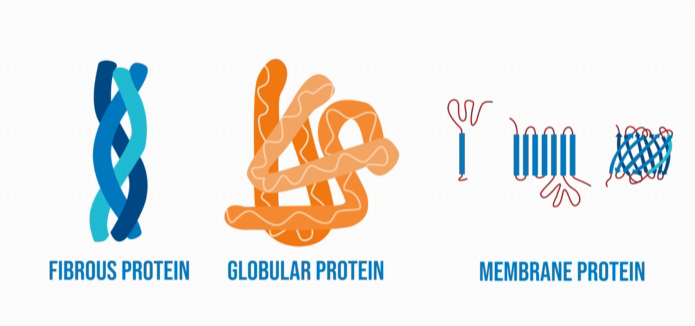
Fibrous proteins are mainly lengthy and narrow chains and are used for structural purposes. They generally have a repetitive sequence of amino acids, and are more durable (or less affected by temperature change, pH change, and so on). Examples of fibrous proteins include: fibrin, actin, myosin, elastin, keratin, and collagen.
Globular proteins are typically more dense and round shaped, and their role is primarily functional. Globular proteins generally have a more irregular sequencing of amino acids, and they are going to be less durable in terms of temperature, and pH changes. Examples of globular proteins include: insulin, immunoglobulin, enzymes, and hemoglobin.
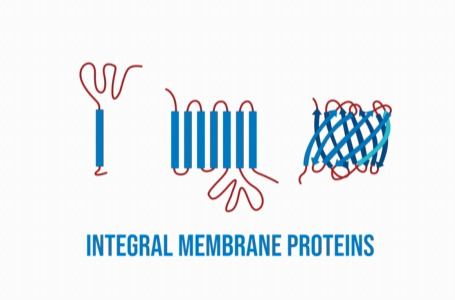
Membrane proteins, like their name suggests, are proteins that are part of biomembranes, or proteins that are interactive with biomembranes. There are two types of membrane proteins: integral membrane proteins, and peripheral membrane proteins. Integral membrane proteins are the proteins that are permanently integrated within the membrane. They are often referred to as intrinsic membrane proteins, because they are located within the core of the membrane. They are able to be permanently attached to the membrane due to their hydrophobic side chains. Most integral proteins spread across the entire membrane.
Peripheral membrane proteins are proteins that are only temporarily apart of a membrane or integral membrane proteins. The way that they attach to the membrane is often indirect, they typically attach by interacting with an integral membrane protein. However, they can attach directly to the membrane, but only by interacting with a lipid polar head. Peripheral membrane proteins are also referred to as extrinsic proteins, because they connect with the exoplasmic face of the membrane.
Function
So, the particular sequence of amino acids in a protein cause it to fold into a three-dimensional shape. Well, the specific shape determines that proteins specific function. A protein has mechanical actions that gets paired with certain chemical occurrences, and this combination of motion and chemistry is what makes the function of proteins so crucial and dynamic. Protein function is dependent on attaching or binding with other molecules. There are specific portions of proteins, and other molecules marked for binding, and these are called binding sites. This allows for proteins to function as catalysts, motors, pumps, signal receptors, or switches. Proteins are the building blocks for our bodies’ tissues, and can also be used as fuel for the body. Protein is found in, effectively, every part of our body.
Source
Protein is found in large amounts in meat, and dairy products. It is also found in beans, nuts, seeds, and vegetables.
Lipids
Structure
Lipids are characterized by their insolubility in water. There are five main classifications of lipids: Fatty acyl(FA); Glycerolipids(GL); Glycerophospholipids(GP); Sterol Lipids(ST); and Sphingolipids(SP).
Fatty acyl refers to fatty acids and their byproducts. It is made up of a hydrocarbon chain on one end and a carboxylic acid group on the other end. The hydrocarbon chain is non-polar and hydrophobic (water fearing). The end containing the carboxylic acid group is polar, and hydrophilic (water loving).
Glycerolipids are also known as triglycerides. Triglycerides are composed of three fatty acids and a glycerol through the process of esterification (the reaction of carboxylic acids and alcohols).
Glycerophospholipids are also known as phospholipids. They are composed of two fatty acids, a glycerol, and a phosphate group. Glycerophospholipids are a major ingredient of cell membranes.
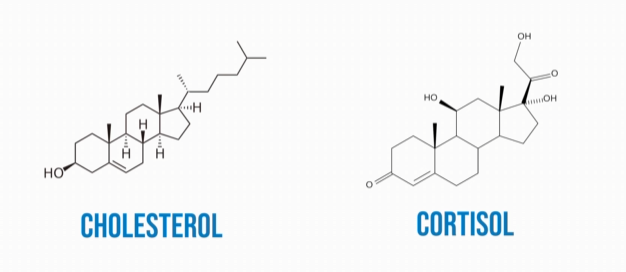
Sterol lipids are a type of steroid with four attached carbon rings and a hydroxyl group. Cholesterol and cortisol are examples of a sterol lipids. Cholesterol helps to form the cell membrane where it serves as secondary messenger in response to extracellular signalling molecules. Cholesterol also helps maintain proper cell fluidity. Cortisol is your body’s primary stress hormone. It is most notorious for aiding your body’s fight-or-flight response.
Sphingolipids are a type of lipid with a foundation made up of sphingoid bases (sphingoid bases contain 2 or 3 hydroxyl groups, and generally a trans-double bond in the 4th position). The most common types of sphingolipids in our bodies are dihydrosphingosine and sphingosine. Sphingolipids are known to protect the surface of a cell from harmful environmental factors; they are also involved in signalling and cellular recognition.
Function
Overall, lipids have crucial functions within cellular processes; this includes storing energy, communication, protection, and they offer structural stability.
Source
Saturated fats are common in animal fats, and are solid at room temperature. Unsaturated fats are common in plants, and are liquid at room temperature.
Nucleic Acids
Structure
Nucleic acids are composed of nucleotides. Nucleotides have three subunits: a nitrogenous base, a 5-carbon sugar, and a phosphate group. There are two types of nucleic acids: Deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). DNA and RNA get their names from the type of sugar they are composed of. DNA contains the sugar deoxyribose, and RNA contains the sugar ribose. There are two types of nitrogenous bases: purine bases and pyrimidine bases. Purine bases have two rings in their structure; adenine and guanine are the purine bases found in both DNA and RNA.
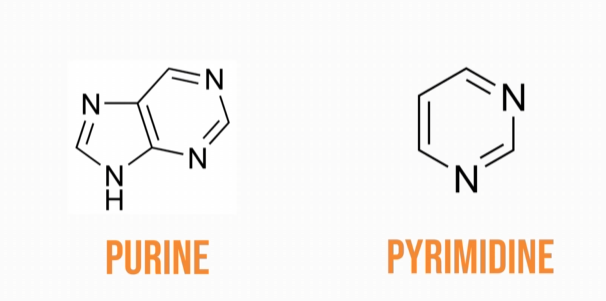
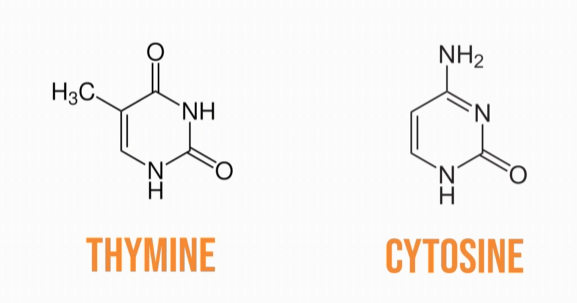
Pyrimidine bases have only one ring in their structure; thymine and cytosine are the pyrimidine bases in DNA,
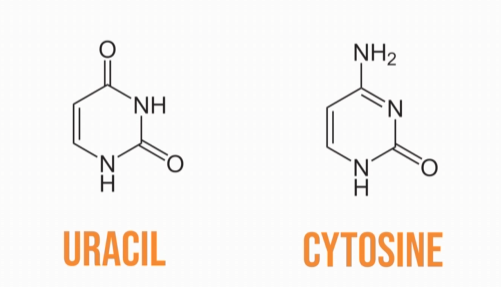
and uracil and cytosine are the pyrimidine bases in RNA.
Function
Nucleic acids make up a cells genetic information. DNA works as an organisms instruction, and RNA works to make a cell’s proteins.
Source
Nucleic acids can be found in vegetables, meat, as well as in yeast.
I hope that this video over macromolecules was helpful.
See you guys next time!
