
Light is a part of our everyday experience and we cannot live without it, but what exactly is light and how does it work? In this video, we’ll discover what light is and see what forms it takes as it interacts with the world around us.
What is light? Although we don’t generally think of the light that we see every day as radiation, that is exactly what light is. Light can be considered electromagnetic radiation and encompasses many forms, including the visible light from light bulbs, the microwaves that cook our food, the x-rays used to make pictures of our insides, and the ultraviolet light that gives us sun burns. All of these are forms of light.
The Wave Nature of Light
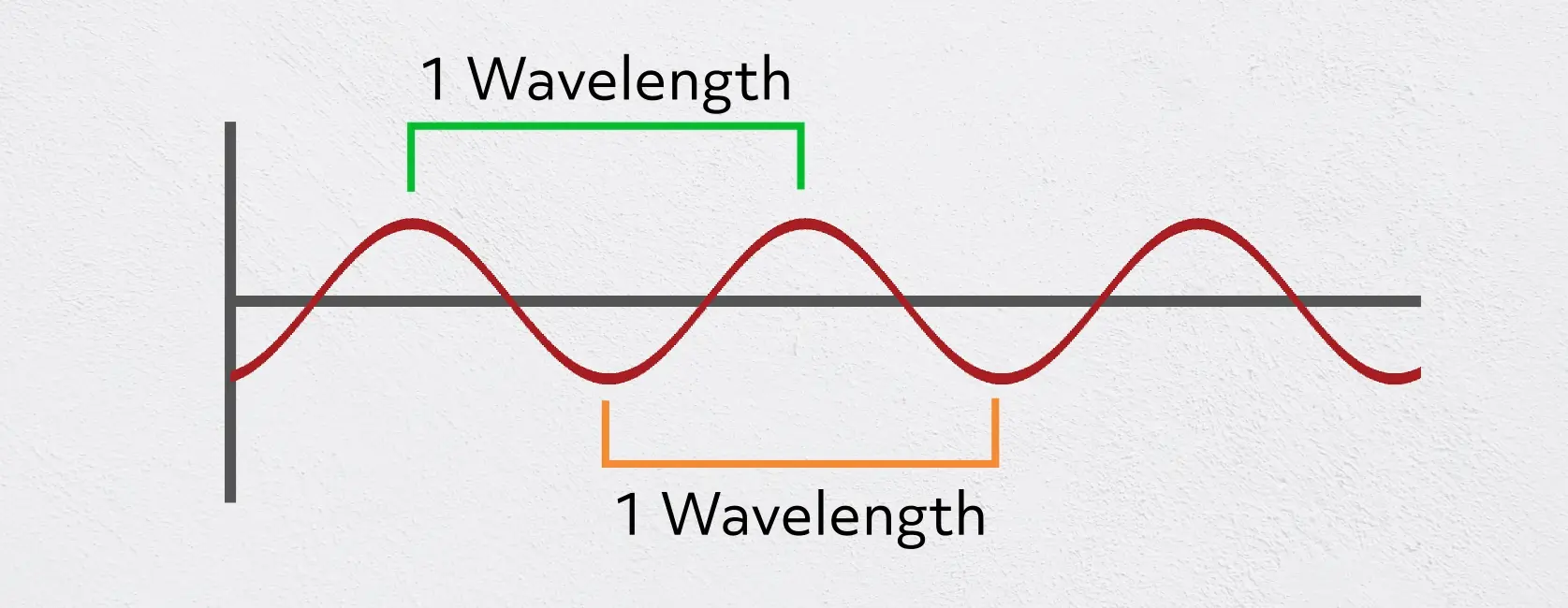
All types of light act as a wave, with a wavelength and a frequency. The wavelength of a light wave is considered to be the distance from crest to crest or trough to trough, where the trough is the lowest part of the wave and the crest is the highest. The frequency can be thought of as the number of oscillations per second or the number of crests or troughs that pass a certain point per second.
In a vacuum, these waves all travel at the speed of light, c, which is roughly 300,000 kilometers per second, faster than anything else in the universe! However, when light is traveling through air, water, or any other materials, it slows down. The amount that it slows down depends on the material.
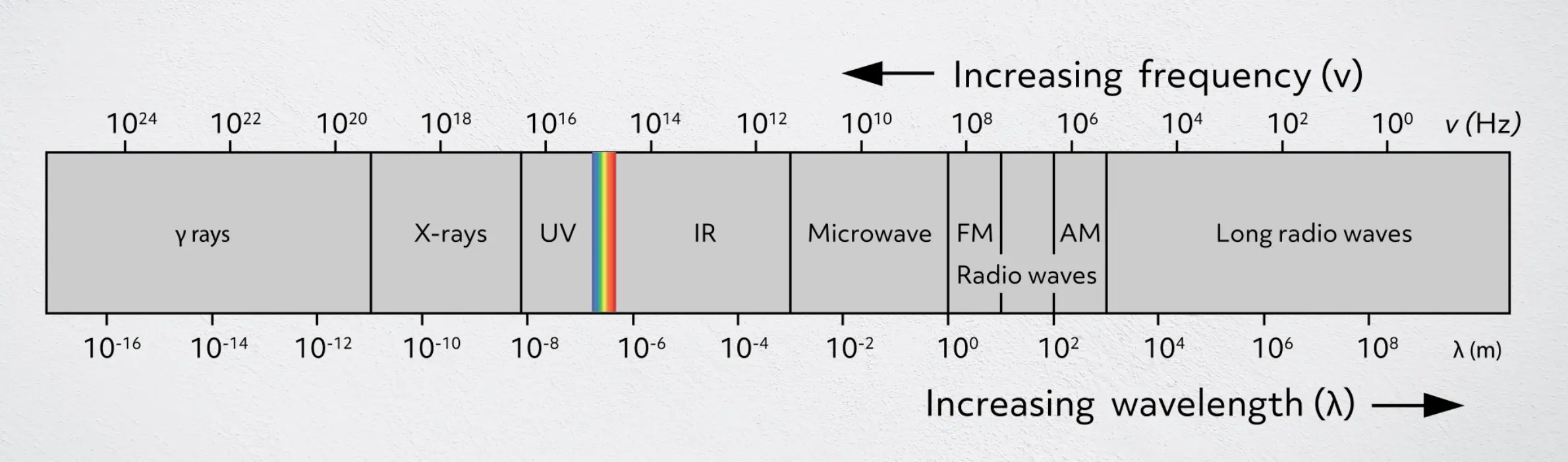
The spectrum of light, as far as scientists can measure, continuously ranges from wavelengths of about 108 meters down to 10-16 meters, with the highest energy light having short wavelengths and high frequencies and the lowest energy light having long wavelengths and low frequencies. This is an incredibly enormous range! The light we are able to see with our eyes is actually only a small range when compared to the entire spectrum. As you can see, the spectrum is categorized into several ranges, from gamma rays all the way to long radio waves.
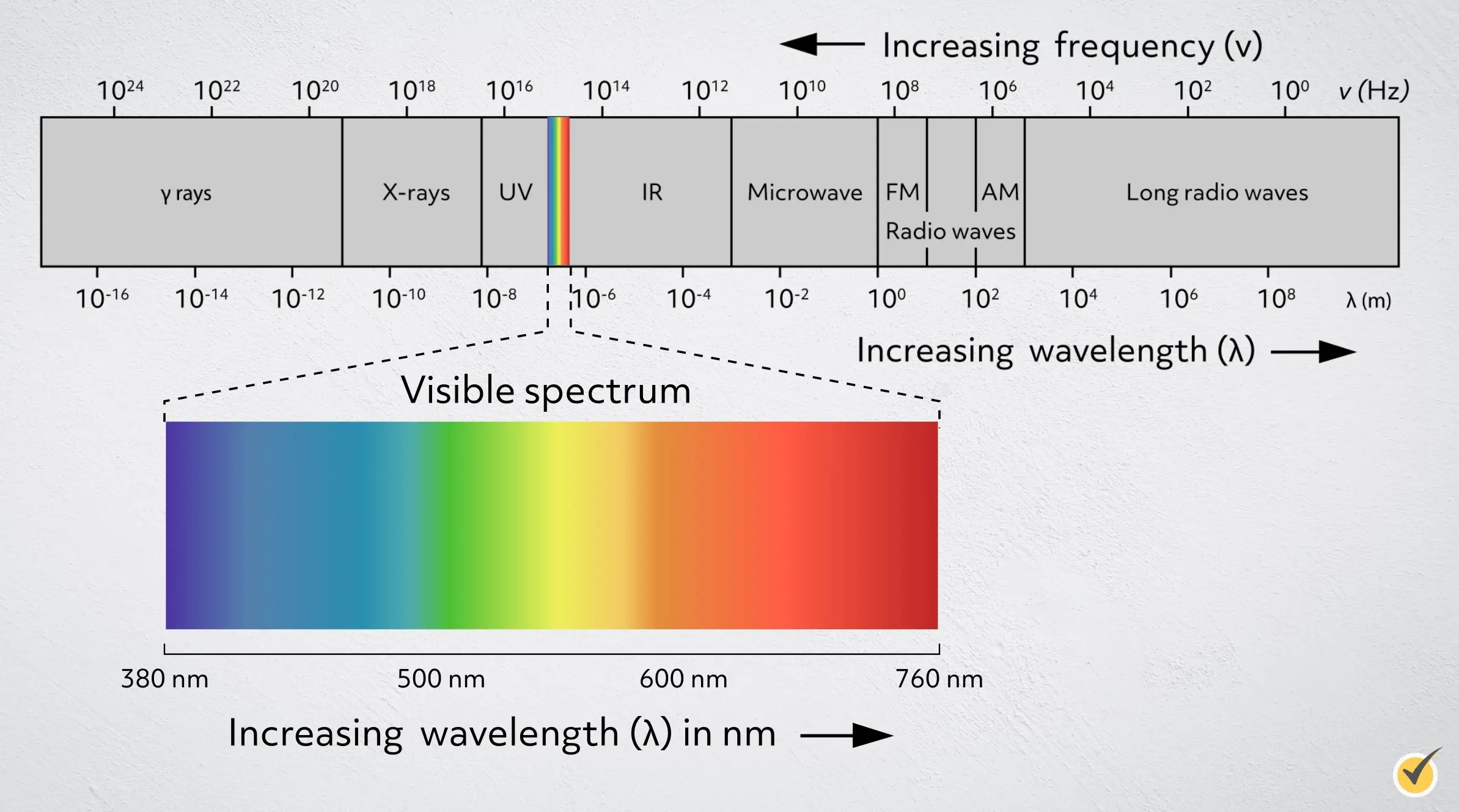
Visible light wavelengths range from about 380-760 nanometers, or 3.8 × 10-7 meters to 7.6 × 10-7 meters. The visible light spectrum is further divided by color, with red light at the long wavelength end and violet at the short wavelength end. We don’t usually see color as individual wavelengths of light in our daily lives, except with special tools like lasers that can isolate a specific wavelength. Instead, we see combinations of wavelengths that our brains process as specific colors. The light from the sun is called “white light” and it contains all of the wavelengths of visible light at approximately equal intensities.
Light Interactions with Matter
Light interacts with matter in various ways, usually depending on the wavelength (or frequency) of the particular light wave and also the composition of the material it is interacting with. When light hits matter, it may be reflected or refracted or it may pass directly through unchanged. When light is reflected, it simply bounces off the reflecting material. When light is refracted, it bends and is then transmitted at a different angle. This generally describes the behavior of light of one wavelength.
But, what if white light hits a refracting surface?
In this case, the varying wavelengths of the light beam cause refraction to happen at different angles for each color of light. This phenomenon is referred to as dispersion. If you’ve ever seen a light going through a prism, you’ve witnessed dispersion.
Prisms, as well as other materials, like glass, have light refracting properties. When white light hits the surface, the different colors of light separate because of their differing wavelengths. Shorter wavelengths (violet) get refracted more than the longer wavelengths (red). They refract again once they leave the prism due to the differences in refractive properties between air and the prism.

Light is an enigmatic thing, in that it can be considered an electromagnetic wave or a particle, called a photon. What we’ve talked about so far are ways that light behaves as a wave. Most waves, like sound waves or waves in water, must have a medium to travel through, but light does not require a medium and is able to travel and spread through a vacuum. This is where the particle properties of light begin to take effect.
In a phenomenon called the photoelectric effect, it was noticed that light that was coming into contact with metal was able to knock electrons off of the surface. In these experiments, light behaves as a particle, because the electrons could only be knocked off or emitted when they received a certain amount of energy from the light. In the classical view of physics, the electrons should have been released after receiving that amount of energy from lower frequency light by storing it until it had enough energy to be emitted. But that wasn’t the case.
The electrons were only emitted when the light they received passed a certain threshold of energy or a higher frequency. It seemed to Einstein that the atoms required specific “packets” of energy in order to emit electrons, which were termed photons. These days, it is generally accepted that light behaves as both waves and particles and that quantum mechanics is to blame for this odd behavior.
Now that we’ve discussed the properties of light, let’s test our knowledge with a couple of questions!
Review Questions
1. Looking at the electromagnetic spectrum, what is the second highest frequency type of electromagnetic radiation of the choices below?
- Blue light
- Yellow light
- X-rays
- AM radio waves
Of the options given the frequencies in decreasing order would be x-rays, blue light, yellow light, and then AM radio waves.
2. Will a prism separate ultraviolet light along with visible light? Would it be refracted more or less than visible light?
- Yes, it will be separated and will be refracted more than all visible light.
- Yes, it will be separated and will be refracted less than all visible light.
- No, a prism will only separate visible colors.
Any type of light can be separated, because they all have different wavelengths and frequencies which will affect the refraction. Since UV light has a higher frequency, it will be refracted more than visible light.
That’s all for our video on light! Thanks for watching, and happy studying!
