
Hi, and welcome to this video on heat transfer. Today, we’re going to focus on heat transfer from a microscopic perspective. In other words, what the molecules, atoms, or particles are doing when heat is transferred.
Reviewing Temperature and Heat
Before we dive into the different types of heat transfer, let’s begin with a review of the technical definitions of temperature and heat. Temperature and heat are words that we use more loosely in everyday language and this video will get confusing if you don’t have a sound grasp on their scientific definitions.
First, temperature is a measurable quantity of a system. For liquids, we simply have to stick a thermometer in, and poof, we’ve got a measurement. On a microscopic level, temperature is related to the average kinetic energy of the particles – the faster and heavier the particles, the higher the temperature.
So if we had two glasses of water, one at 41°F and one at 203°F, the water molecules in the cold glass are moving very slowly and the molecules in the hot glass are moving very quickly.
On the other hand, when scientists measure heat, they don’t measure the heat of a single system. Heat, by scientific definition, is the transfer of thermal energy between systems. So instead of measuring the heat of each water glass, we measure the heat exchanged between each glass and the surroundings.
Some scientists might still take issue with that phrasing, preferring to say we measure the thermal energy exchanged between each glass and the surroundings. But unless you find yourself in a room full of overly pedantic scientists, it’s fine to say heat transfer rather than thermal energy transfer. Today, we’ll use the phrases interchangeably.
Now, let’s move on to the different types of heat transfer: conduction, convection, and radiation. We’ll go through each type from a microscopic perspective and provide some examples. Hopefully, by the end of the video, you’ll be able to identify each type in your surroundings.
Conduction
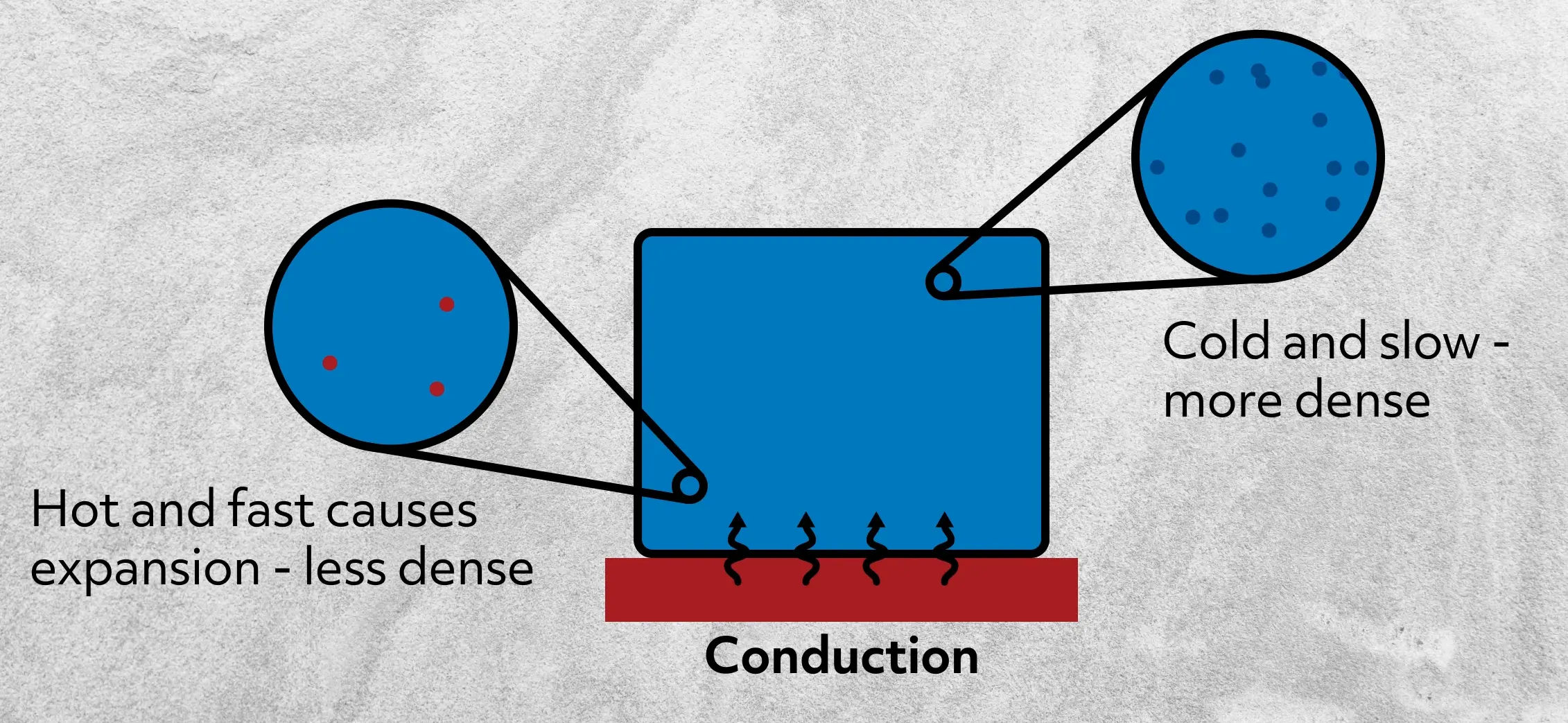
First, conduction is the transfer of thermal energy within a material or between materials in contact.
On a microscopic scale, conduction happens when hot, high-energy particles bump into cold, low-energy particles and transfer some kinetic energy. This increases the energy and temperature of the low-energy particles and decreases the energy and temperature of the high-energy particles. Conduction continues until the system reaches thermal equilibrium, in other words, when the particles are all at the same temperature.
This means that hot particles always heat up cold particles. In other words, thermal energy flows from the high-energy to the low-energy particles. For instance, when you hold an ice cube in your hand, you can feel the cold. It’s easy to think, “Oh, the cold is entering my hand.” But the more accurate way to think about that scenario is that heat is traveling out of your hand into the ice cube, which lowers the heat in your hand, making your hand colder.
Of course, different materials are better conductors of thermal energy than others. Solids and liquids are generally better than gases because so many more particles are in contact with each other, thus facilitating a quick transfer of energy.
So, for instance, if you needed to cool down a cup of tea, it would happen much faster in a bowl of water at 36°F rather than in your fridge, even if it’s also at 36°F.
That’s because in the bowl of water, the outside of your mug is in contact with trillions and trillions of water molecules, which readily offer a place for heat to be transferred. In the air, there are significantly fewer particles to receive that thermal energy, so it will take a much longer time to transfer the same amount of heat.
Convection: Fluid Movement
Alright, let’s move onto convection, the transfer of heat through the bulk movement of fluids. In physics, a fluid is simply a substance that continually flows under an external force; this includes both liquids and gases. Convection can either be natural or forced- as in, it can happen due to natural phenomena, like gravity or buoyancy, or because of an external force, like a pump or fan.
Let’s start with an example of natural convection.
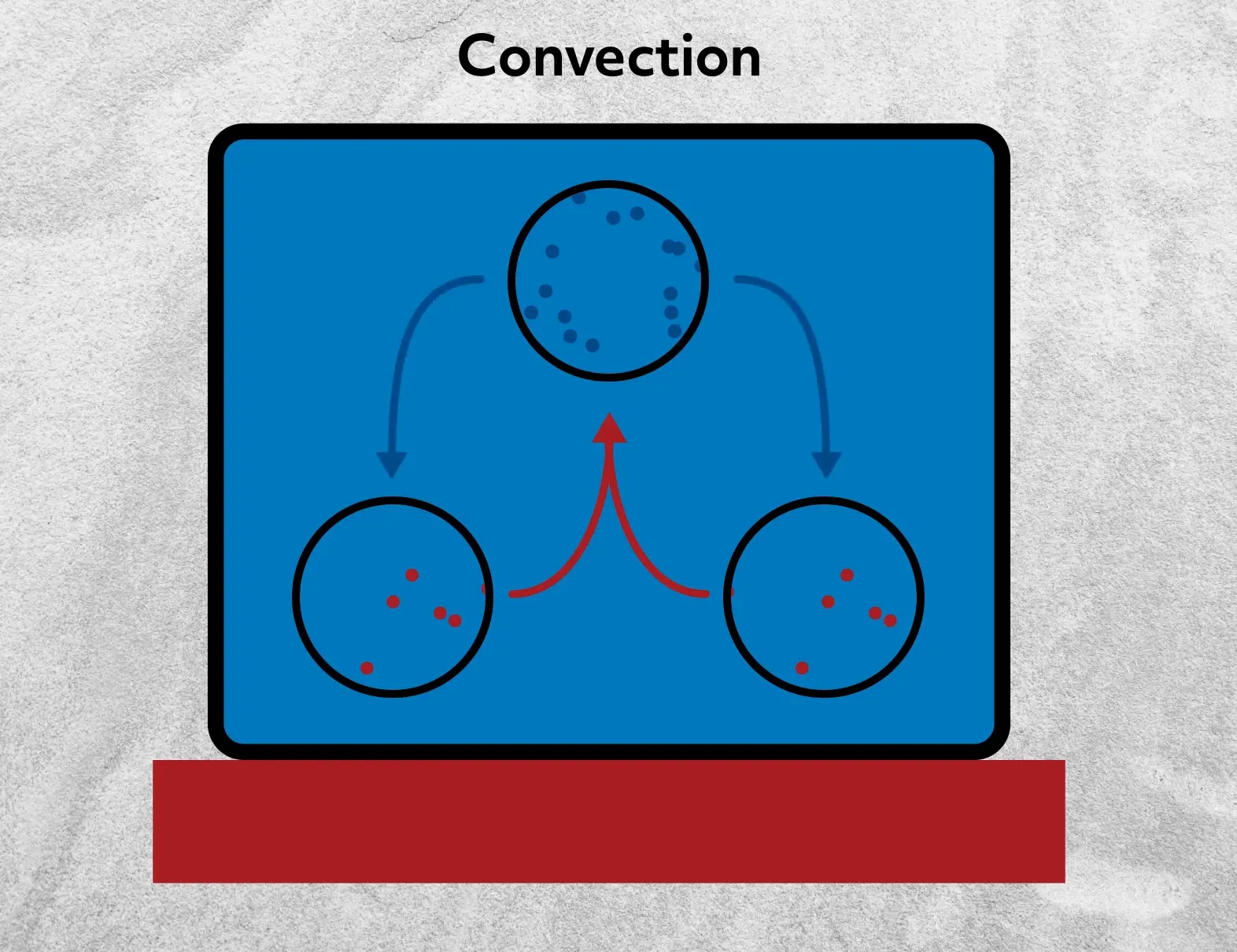
Consider a pot of water on an electric stovetop. Conduction is responsible for the initial transfer of thermal energy to the bottom of the pot, heating up the lower portion of water. That water expands as it is heated and is less dense than the cold water above it. We can understand this from a molecular perspective, consistent with what we’ve already discussed today about temperature. The hot water molecules have a lot of kinetic energy and require more space to move around in comparison to the cold water molecules.
We now have less dense water at the bottom of the pot, so it will rise due to buoyancy while the colder, denser water will sink. Subsequently, that cold water, now closer to the heat source, will become heated and eventually be the more buoyant water and will rise in turn. This continuous movement results in currents in the water where heat is being transferred throughout the whole pot and not through conduction alone, though conduction is still happening on the stove and between individual water molecules.
While this is a pretty simplified explanation of the fluid mechanics involved, it does cover the general concept of natural convection.
Now, let’s consider forced convection, which you probably interact with on a regular basis. Hair dryers, central heating systems in buildings, and convection ovens are all examples of forced convection. Basically, rather than relying on a natural process, we employ a fan or pump to move the fluid around, thus transporting thermal energy throughout the system.
Radiation
Lastly, let’s talk about thermal radiation.
All matter, provided that it is warmer than absolute zero, emits thermal radiation. And by radiation, I mean electromagnetic waves.
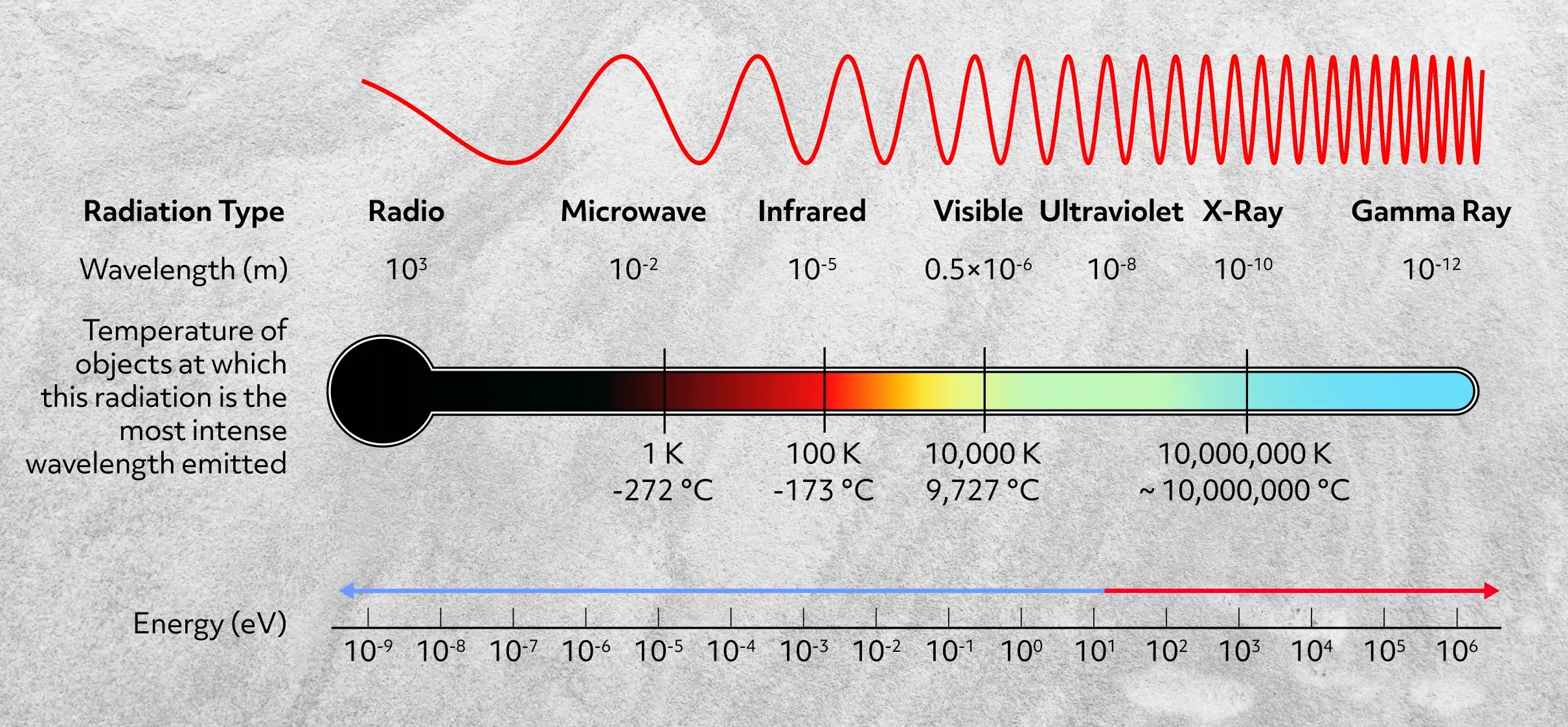
If you’re not familiar with the electromagnetic spectrum, here’s a look, showing the relationship between wavelength and energy.
The shorter the wavelength, the higher the energy. And as the name suggests, the waves are oscillating electric and magnetic fields, which do not require a medium to move through. In other words, they can travel through a vacuum at the speed of light.
Substances emit different wavelengths of radiation depending on their temperature. The hotter the substance, the shorter the wavelength of the thermal radiation. The cooler the substance, the longer the wavelength. This results from random physical motions of particles, which cause charge acceleration and dipole oscillations, which in turn release electromagnetic radiation.
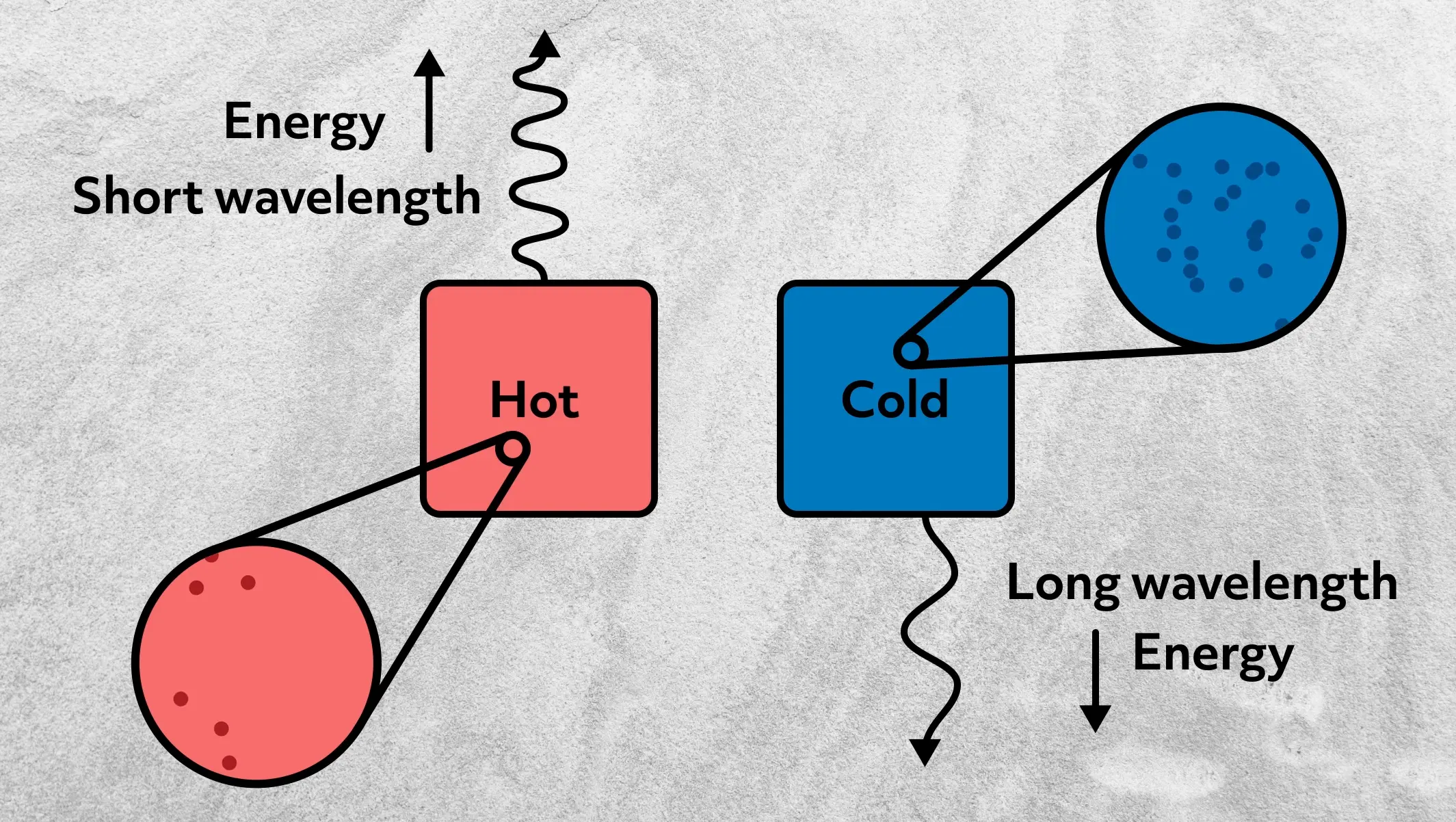
Thus, we can measure temperature by recording the wavelength of the thermal radiation being released. This is actually the principle behind night-vision devices. Animals and humans are about 310 K, which results in the constant release of infrared radiation. Night-vision devices capture the infrared radiation and convert it into a visible image.
The sun, on the other hand, which is 5800 K, releases very high-energy radiation, like ultraviolet and visible light. Another example that many of you are probably familiar with are the heating coils on electric stoves. When turned to high, they glow red because they are so hot (755 K) that they begin to release visible red light.
Thus far, we’ve only talked about the emission of thermal radiation, but radiation can also be absorbed and converted into kinetic energy.
For example, the Sun’s thermal radiation passes through our atmosphere and is absorbed by the Earth.
This heats the Earth, which in turn releases its own thermal radiation, but at much longer wavelengths than the Sun, because, of course, the Earth is much cooler than the Sun. The Earth, which is effectively 255 K, releases infrared radiation. That infrared radiation is then captured by atmospheric gases, which causes our atmosphere to warm. You might have noticed that, unlike conduction and convection, radiation doesn’t require contact between substances.
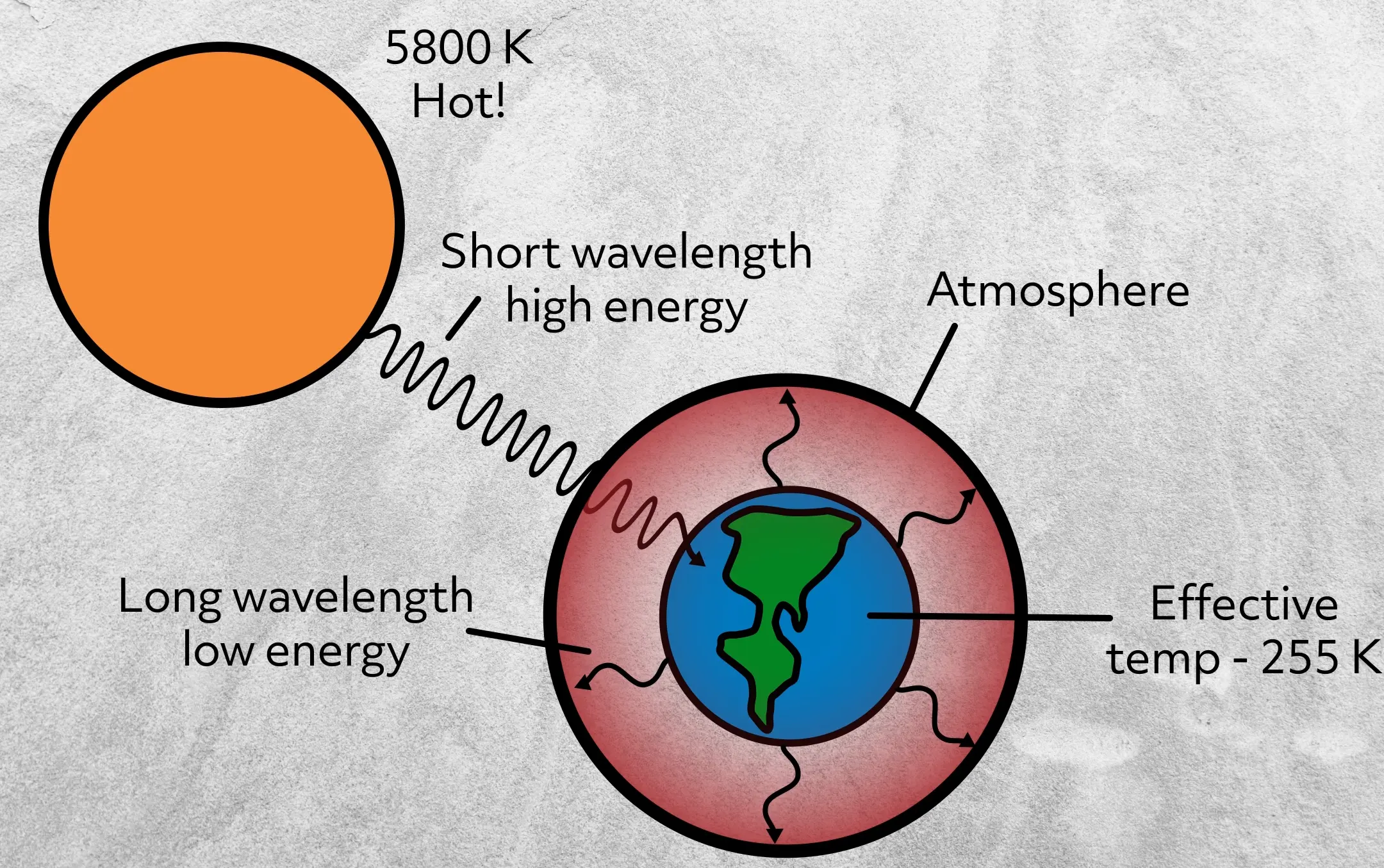
Note that while radiation results in the transfer of heat, it doesn’t do so until it is absorbed. Once absorbed, the energy can be converted in a number of ways, one of them being heat. Remember, space is cold even close to the sun because there’s no matter to absorb the radiation and convert it to thermal energy.
It’s important to note that atmospheric gases don’t absorb much ultraviolet or visible light coming from the Sun, but they do absorb infrared radiation from the Earth. What scientists learned from observing and measuring this phenomenon is that materials absorb specific wavelengths and reflect or transmit other wavelengths. We need quantum mechanics to get into a detailed explanation on how this works, which is beyond this video, but for now, just be aware that not all materials absorb all radiation.
Alright, let’s wrap up with a review and a little practice.
We covered the three different types of heat transfer: conduction, convection, and radiation. Conduction is the direct transfer of heat from hot, high-energy particles to cold, low-energy particles. Convection is the transfer of heat through bulk movement of fluids, either naturally or forced. And lastly, radiation is the transfer of heat through electromagnetic waves.
Let’s finish with two exercises. First, let’s look at a convection oven and identify each different type of heat transfer.
There are metal coils that are hot enough to emit significant and high-energy thermal radiation, which then heats the surface of the food or the container. Then, conduction takes over and heat is transferred into the center of the food. Lastly, a convection oven has a fan to continuously and evenly circulate the hot air in the oven.
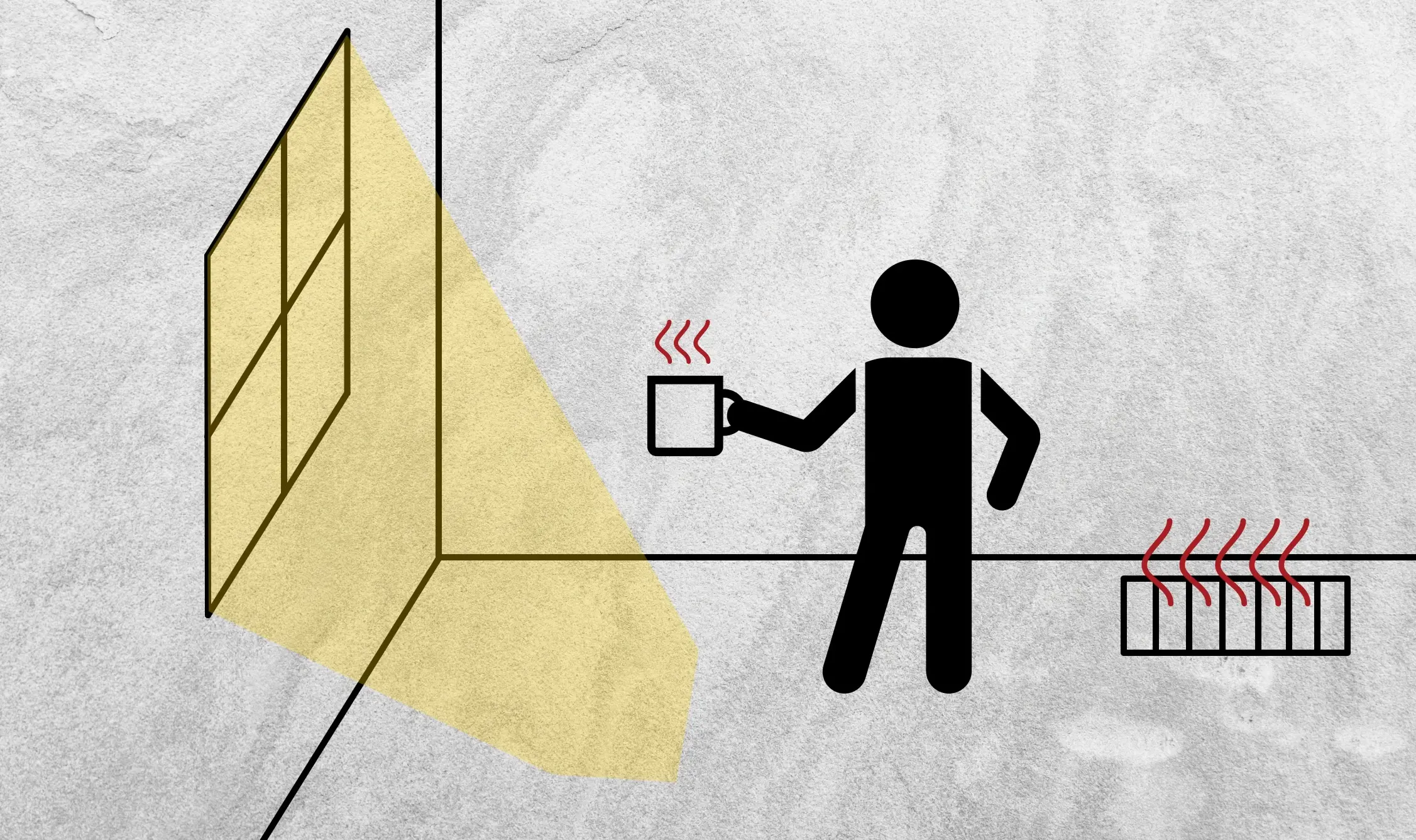
Secondly, take a minute to observe this picture. Can you spot the three types of heat transfer happening here? For radiation, notice that the sunlight is coming in from the window, heating a patch of the carpet. If you take a look at the floor vent, you’ll notice that hot air is blowing into the room, which would be forced convection.
And lastly, the transfer of thermal energy from the hot coffee mug into the person’s hand is an example of conduction. Basically, heat transfer is happening all around you, you just have to take note.
Alright, that’s it for this overview of heat transfer! Thanks for watching, and happy studying!
